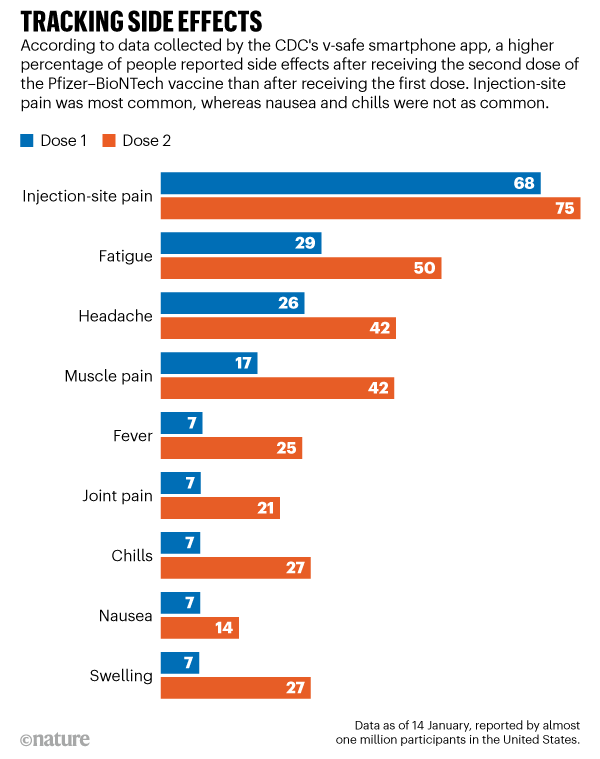
 Reporting rates of adverse events following COVID-19 vaccination, including those for children and adolescents, are very stable. The TGA is closely monitoring adverse event reports in people aged under 18 years. The most up-to-date vaccine recommendations for children are available from ATAGI. Vaccine safety in children and adolescents These are recognised side effects of vaccination and are usually transient and mild. Skin reactions at the site of the injection are also common and can include pain, swelling, redness and an itchy rash. The most frequently reported side effects suspected to be associated with the vaccines include headache, muscle and joint pain, fever, chills and nausea. Learn more about how to report a suspected side effect to a COVID-19 vaccine. by calling 1300 Medicine (1300 633 424) or visiting You can report anonymously.Learn more about how the TGA identifies and responds to safety issues.Īnyone can report a suspected side effect, either: The Vaxzevria (AstraZeneca) vaccine is no longer available in Australia. Detailed information of our analysis of thrombosis with thrombocytopenia syndrome (TTS), Guillain-Barre syndrome (GBS) and immune thrombocytopenia (ITP) following Vaxzevria (AstraZeneca) in adults is available in a previous vaccine safety report.It occurs in males and females but is more common after the second dose in boys aged 12-17 years (13 cases per 100,000 Comirnaty doses and 24 cases per 100,000 Spikevax doses) and men under 30 (9 cases per 100,000 Comirnaty doses and 20 cases per 100,000 Spikevax doses). It is reported in around 1-2 in every 100,000 people who receive Comirnaty (Pfizer) and around 2 in every 100,000 of those who receive Spikevax (Moderna). Myocarditis is a known but very rare side effect of the mRNA vaccines.These are usually temporary conditions, with most people getting better within a few days. We are carefully monitoring and reviewing reports of myocarditis and pericarditis (inflammation of the heart or membrane around the heart) following vaccination.More detail on vaccine safety in children, adolescents and adults following vaccination is available in a previous vaccine safety report. Reporting rates of adverse events following COVID-19 vaccination are very stable.The most up-to-date recommendations for use of the COVID-19 vaccines are available from the Australian Technical Advisory Group on Immunisation (ATAGI).This reflects what was seen in the clinical trials. 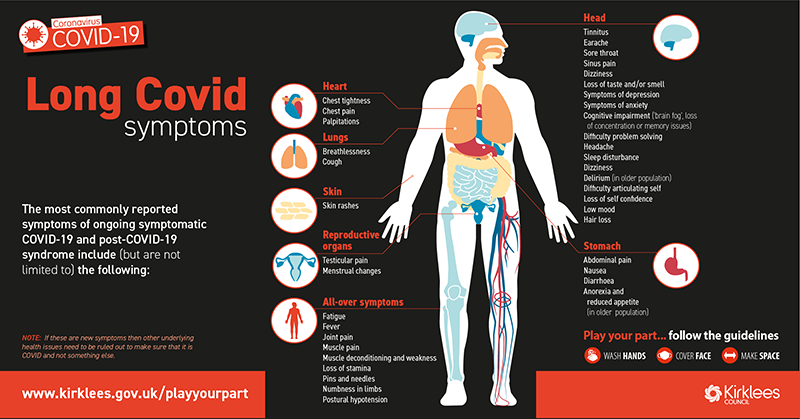 /cloudfront-us-east-1.images.arcpublishing.com/gray/QJLMYNB4J5EQHEKZ2RNSAJLWQM.jpg)
The most frequently reported include injection-site reactions (such as a sore arm) and more general symptoms, like headache, muscle pain, fever and chills.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
